Hello students and welcome to our solutions on Class 7 Science Curiosity Chapter 5! The most important and fundamental parts of this chapter are the concepts on physical and chemical, reversible and irreversible changes. We suggest that you start with these basic concepts and then delve into the rest of the chapter. Please access the solutions below!
Happy studying!
Let Us Enhance Our Learning (Class 7 Science Curiosity Chapter 5):
1. Which of the following statements are the characteristics of a physical change?
(i) The state of the substance may or may not change.
(ii) A substance with different properties is formed.
(iii) No new substance is formed.
(iv) The substance undergoes a chemical reaction.
(a) (i) and (ii) (b) (ii) and (iii)
(c) (i) and (iii) (d) (iii) and (iv)
Answer: (c) (i) and (iii)
In a physical change only physical properties like shape, size, and state of substances change and no new substance is formed.
2. Predict which of the following changes can be reversed and which cannot be reversed. If you are not sure, you may write that down. Why are you not sure about these?
(i) Stitching cloth to a shirt
(ii) Twisting of straight string
(iii) Making idlis from a batter
(iv) Dissolving sugar in water
(v) Drawing water from a well
(vi) Ripening of fruits
(vii) Boiling water in an open pan
(viii) Rolling up a mat
(ix) Grinding wheat grains to flour
(x) Forming of soil from rocks
Answer:
(i) Irreversible change. When you stitch a cloth to form a shirt, you alter it permanently.
(ii) Reversible change. The straight string can be twisted and straightened again.
(iii) Irreversible change. An irreversible chemical reaction happens during cooking.
(iv) Reversible change. You can evaporate the water to get the sugar back.
(v) Reversible change. You can pour the water back into the well.
(vi) Irreversible change. Ripening of fruits is an irreversible change because chemical reaction takes place to form new products.
(vii) Reversible change. The evaporated water can be recovered by condensation.
(viii) Reversible change. The mat can be straightened again back to its original position.
(ix) Irreversible change. It is practically impossible to get wheat grains back from flour.
(x) Irreversible change. Rocks undergo physical and chemical changes due to weathering to form soil.
3. State whether the following statements are True or False. In case a statement is False, write the correct statement.
(i) Melting of wax is necessary for burning a candle. (True/False)
(ii) Collecting water vapour by condensing involves a chemical change. (True/False)
(iii) The process of converting leaves into compost is a chemical change. (True/False)
(iv) Mixing baking soda with lemon juice is a chemical change. (True/False)
Answer:
(i) True. The wax of the candle melts, is carried up the wick, and evaporates due to the heat of the flame.
(ii) False. Collecting water vapour by condensing is a physical change because it just involves a change of state.
(iii) True. The process of decomposing leaves into compost involves chemical reactions, which is irreversible.
(iv) True. When baking soda is mixed with lemon juice, new substances such as carbon dioxide is produced. Thus, it is a chemical change.
4. Fill in the blanks in the following statements:
(i) Nalini observed that the handle of her cycle has got brown deposits. The brown deposits are due to ________, and this is a________ change.
(ii) Folding a handkerchief is a _________________ change and can be________.
(iii) A chemical process in which a substance reacts with oxygen with evolution of heat is called _________, and this is a __________ change.
(iv) Magnesium, when burnt in air, produces a substance called __________. The substance formed is _________ in nature. Burning of magnesium is a _________ change.
Answer:
(i) Nalini observed that the handle of her cycle has got brown deposits. The brown deposits are due to rust, and this is a chemical change.
(ii) Folding a handkerchief is a physical change and can be reversed.
(iii) A chemical process in which a substance reacts with oxygen with evolution of heat is called combustion, and this is a chemical change.
(iv) Magnesium, when burnt in air, produces a substance called magnesium oxide. The substance formed is basic in nature. Burning of magnesium is a chemical change.
5. Are the changes of water to ice and water to steam, physical or chemical? Explain.
Answer:
The changes of water to ice and water to steam are physical changes. This is because only the state of the substances changes when water freezes to form ice and boils to form steam. No new substances is formed and the changes are easily reversible.
6. Is curdling of milk a physical or chemical change? Justify your statement.
Answer:
Curdling of milk is a chemical change because a chemical reaction happens in which milk forms a new substance curd with sour taste and different chemical composition.
7. Natural factors, such as wind, rain, etc., help in the formation of soil from rocks. Is this change physical or chemical and why?
Answer:
The formation of soil from rocks involves both physical and chemical changes.
The change caused by natural factors like wind, rain, and temperature fluctuations in the formation of soil from rocks is both physical and chemical. Physical changes occur when rocks break into smaller pieces due to processes like freezing and thawing of water or root expansion, without altering their composition. Chemical changes happen when water or air reacts with minerals in the rocks, forming new substances, such as iron in basalt reacting with water and oxygen to form iron oxide. Thus, soil formation involves a combination of both physical and chemical weathering.
8. Read the following story titled ‘Eco-friendly Prithvi’, and tick the most appropriate option(s) given in the brackets. Provide a suitable title of your choice for the story.
Prithvi is preparing a meal in the kitchen. He chops vegetables, peels potatoes, and cuts fruits (physical changes/chemical changes). He collects the seeds, fruits, and vegetable peels into a clay pot (physical change/chemical change). The fruits, vegetable peels, and other materials begin to decompose due to the action of bacteria and fungi, forming compost (physical change/chemical change). He decides to plant seeds in the compost and water them regularly. After a few days, he notices that the seeds begin to germinate and small plants start to grow, eventually blooming into colourful flowers (physical change/chemical change). His efforts are appreciated by all his family members.
Answer:
Prithvi is preparing a meal in the kitchen. He chops vegetables, peels potatoes, and cuts fruits (physical changes). He collects the seeds, fruits, and vegetable peels into a clay pot (physical change). The fruits, vegetable peels, and other materials begin to decompose due to the action of bacteria and fungi, forming compost (chemical change). He decides to plant seeds in the compost and water them regularly. After a few days, he notices that the seeds begin to germinate and small plants start to grow, eventually blooming into colourful flowers (chemical change). His efforts are appreciated by all his family members.
A suitable title for the story would be, “Prithvi’s Green Kitchen.”
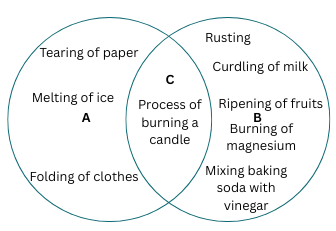
9. Some changes are given here. Write physical changes in the area marked ‘A’ and chemical changes in the area marked ‘B’. Enter the changes which are both physical and chemical in the area marked ‘C’.
Process of burning a candle; Tearing of paper; Rusting; Curdling of milk; Ripening of fruits; Melting of ice; Folding of clothes; Burning of magnesium and Mixing baking soda with vinegar.
Answer:
10. The experiments shown in Fig. 5.11a, b, c, and d were performed. Find out in which case(s) did lime water turn milky and why?
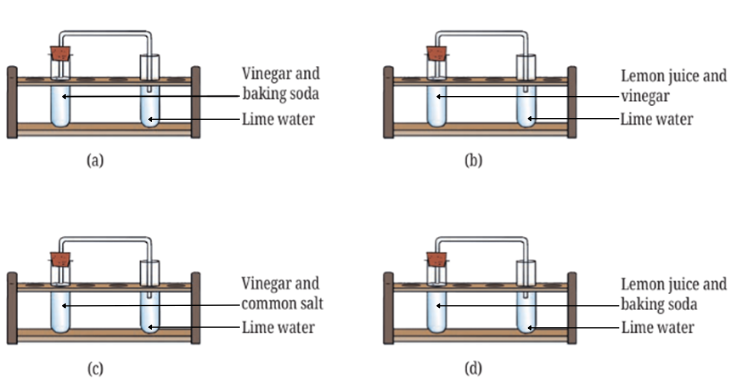
Answer: (a) and (d)
In (a) acidic vinegar and basic baking soda react to form carbon dioxide gas which turns lime water milky. In (d) acidic lemon juice and basic baking soda react to form carbon dioxide gas which turns lime water milky as well.
In (b) lemon juice and vinegar are both acidic, hence they do not react to form CO2.
In (c) acidic vinegar will not react with common salt to produce CO2.
Hope you enjoyed our solutions material on Class 7 Science Curiosity Chapter 5! Click on the links below to access the solutions to the remaining chapters!
Related Links:
Chapter 1 The Ever-Evolving World of Science
Chapter 2 Exploring Substances: Acidic, Basic, and Neutral
Chapter 3 Electricity: Circuits and their Components
Chapter 4 The World of Metals and Non-metals
Chapter 5 Changes Around Us: Physical and Chemical
Chapter 6 Adolescence: A Stage of Growth and Change
Chapter 7 Heat Transfer in Nature
Chapter 8 Measurement of Time and Motion
Chapter 9 Life Processes in Animals
Chapter 10 Life Processes in Plants
Chapter 11 Light: Shadows and Reflections
Chapter 12 Earth, Moon, and the Sun


